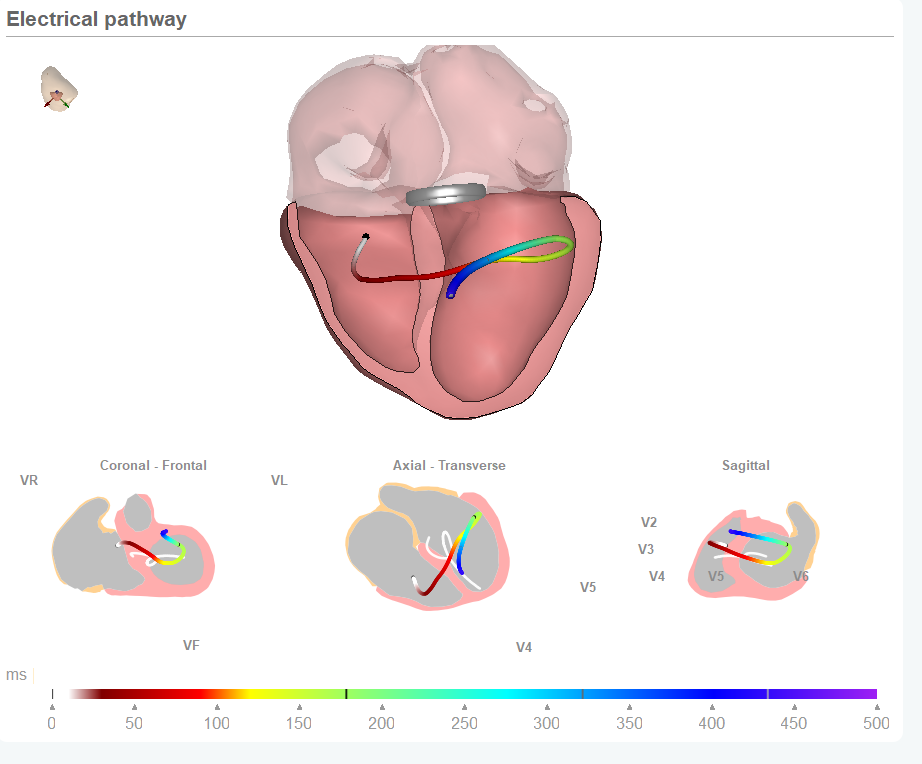
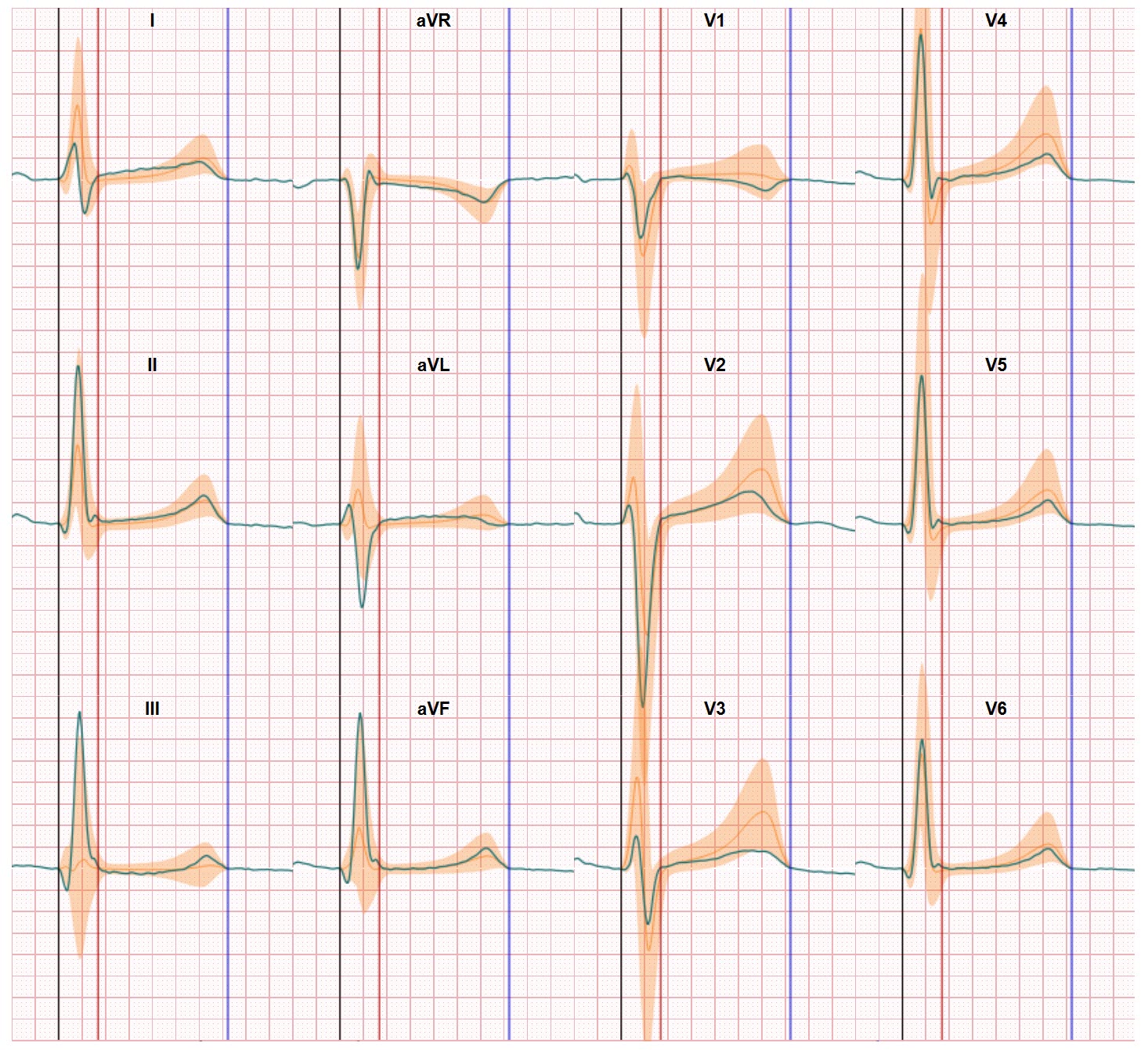
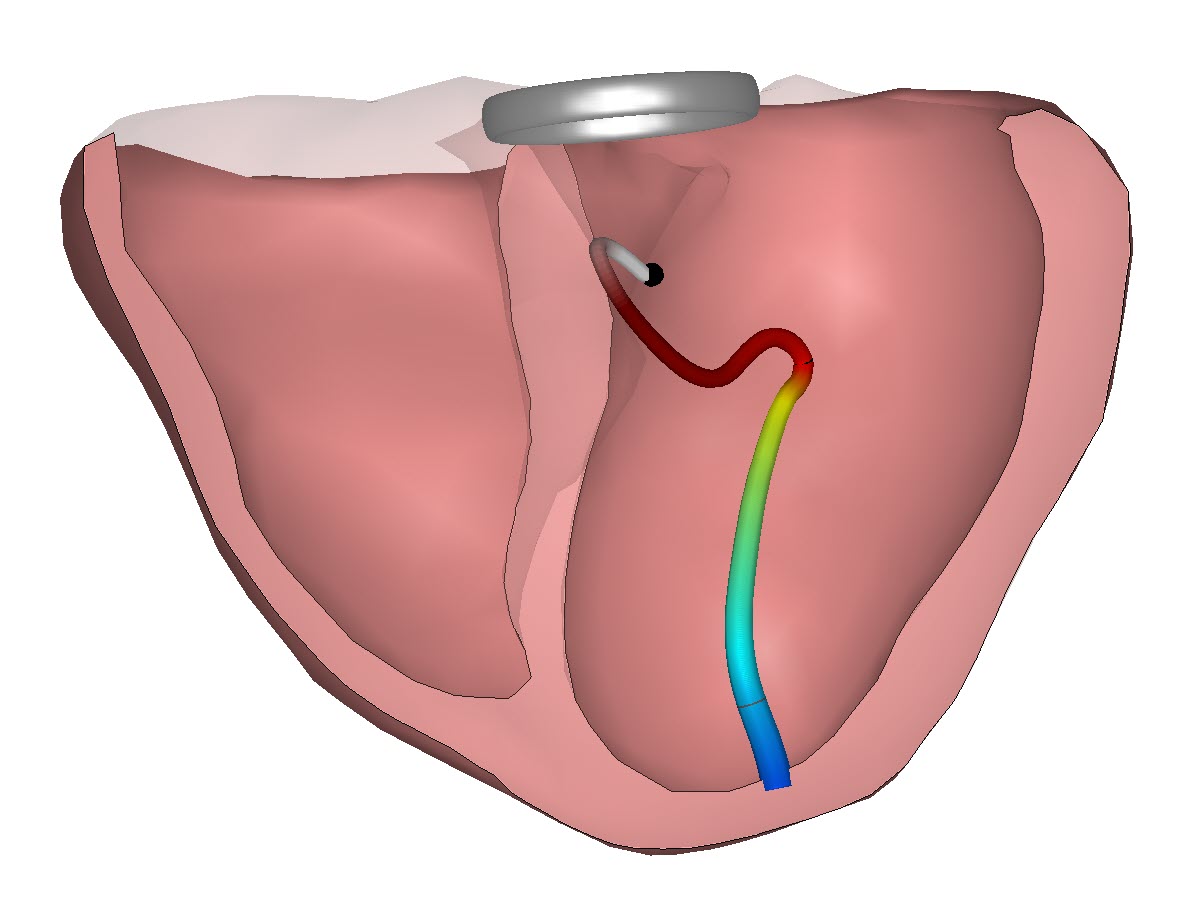
Trusting digital health solutions is one of the key challenges which need to be solved before the medical professionals will fully embrace the options offered in the market. In the World Heart Foundation digital health roadmap report we discussed last week in our Blog it showed the that a vast majority of cardiovascular medical professionals see the (perceived) quality of these digital health solutions as a major barrier. And not without reason. Recently the WHO reported that only 7% of these solutions are properly evaluated before they are brought to market. And a quick look across the many solutions offered shows that the majority can only be seen as ‘gadgets’. This is not all the ‘fault’ of the manufacturers of these (virtual) devices. We understand many of them have embarked on a development route with the intention to build something proper and robust. But then their development and validation processes are confronted with a void in evaluation standards. And this absence of evaluation standards hinders the effectiveness evaluation by both physicians, patients, payers and regulators.
And Artificial Intelligence based digital solutions are confronted with the lack of available data to both train the neural networks and validate the results. And furthermore, even if data is available there’s always the risk of existing structural bias in the data. Running Real Clinical Trials to collect the enormous amounts of data you need to train and validate AI solutions for cardiovascular digital solutions is the next big hurdle.
Gaining trust in digital health solutions
But despite all this barriers the need for better, faster, more accurate digital heart health solutions still drives innovation. And the WHF report already shows optimistic signs of changing rules on evidence reporting and assessment. Therefore we see the journey to trusting digital health solutions has really started. We see the availability of qualified data sets growing slowly although some of these sets are behind thick paywalls. And the FDA is active in advising subsequent post-market access periodic updates which are necessary to monitor real-world performance of the digital solutions.
As a team involved in developing breakout innovative digital solutions in Electrocardiogram interpretation and ECG based diagnostics we keep a sharp eye on these developments. But the current steeple chase race to acquire market acceptance and even market entry has also influenced our technology approach. Interestingly our current ECG interpretation has actually benefited from the barriers. A major breakout in ECG interpretation performance can already be achieved by applying more classical technologies such as statistical analysis and data standardization of the Electrocardiogram. And this technology also counts as a digital heart health solutions which creates benefits for both patients and medical professionals.